Enartis News – Red Wines: how to prevent post-bottling defects
This second document on post-bottling defects is dedicated to red wines. Many of the potential defects that can appear in bottled red wine can also be found in white wines and have been described in part one of this newsletter series. For this reason, we’ll focus on a problem specific to reds: color and tannin precipitation.
COLOR INSTABILITY
Appearance: dark red amorphous sediment mainly formed by anthocyanins, tannins and polysaccharides in young wines. Potassium bitartrate crystals can be found as well, precipitated as a result of color colloids sedimentation.

In old wines, dark layers coating the side of the bottle, mainly from tannins and anthocyanins can be seen.
Causes: red wines exposure to low temperatures can cause the precipitation of colloidal color matter.
In young wines, colloidal complexes are made of anthocyanins, tannins and polysaccharides. These compounds tend to polymerize and pass from the soluble state to the colloidal state by forming large aggregates that in time precipitate and form a
sediment on the bottom of the bottle. Polymerization is faster in summer, favored by high temperatures, while color precipitation happens more frequently during winter, favored by low temperatures. Wines rich in colloidal color matter and more prone to
color precipitation are wines produced from moldy grapes, high-temperature fermentations or strong mechanical action (rough crushing, pumping, excessive pump-over, stirring lees, etc.).
In old red wine, colloidal color matter precipitate is mainly caused by tannins that polymerize by oxidative condensation. The process takes time, depending on the quantity of oxygen that can permeate through the cork. Exposure to cold temperatures contribute to color sedimentation.
Color Stability Test
To check wine color stability, running a simple test is recommended:
a) Filter 100 mL of wine on a 0.45 microns membrane.
b) Measure turbidity (T1) with a nephelometer (turbidimeter).
c) Place 100 mL of wine in the drop shaped flask (picture 2) or in a 125 mL clear glass bottle.
d) Place the sample in the freezer at -4°C.
e) After 24 hours, check for the presence/absence of sediment. Presence of an amorphous dark red sediment indicates that wine
is color unstable.
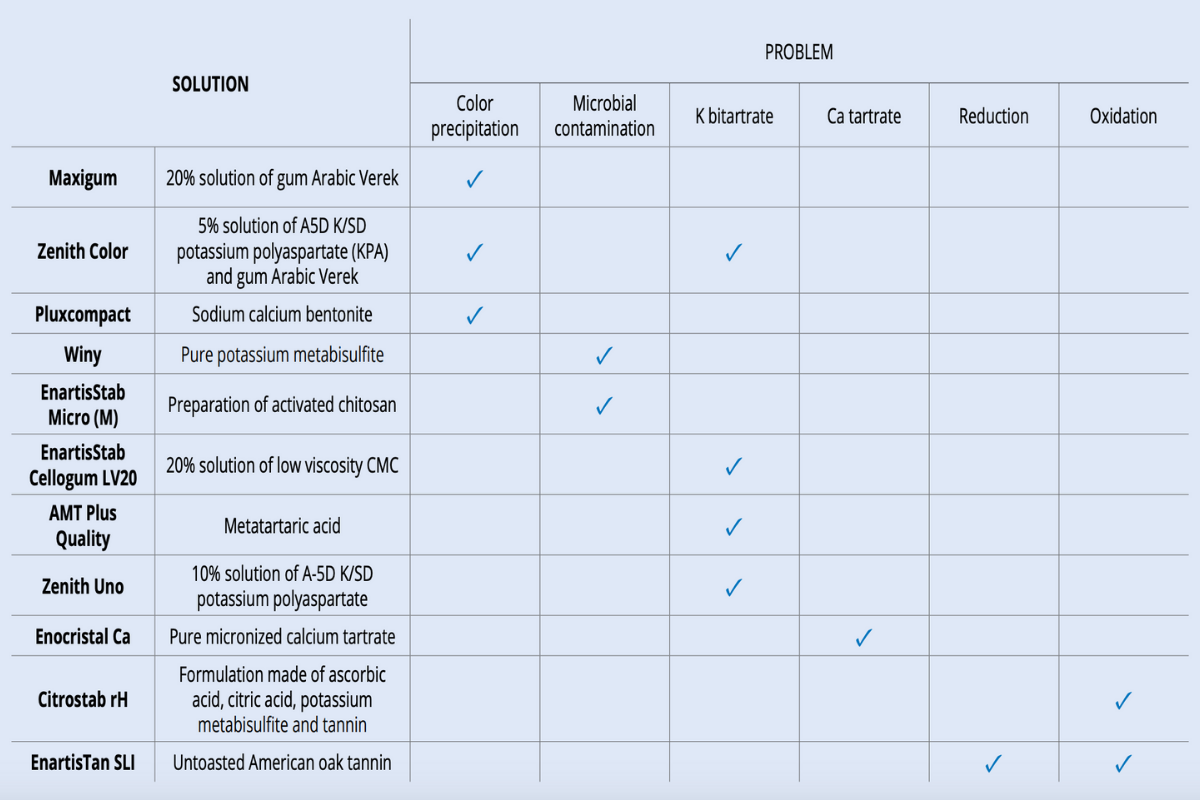
Prevention: There are two methods to avoid color precipitation:
1) Addition of gum Arabic
Gum Arabic’s protective effect is attributed to a coating of the colloid particles that prevents them from agglomerating. In fact, gum is adsorbed by the colloidal color particles and then its hydrophilic part spreads in the solution maintaining a separation between the various colloidal particles. Stability is assured when there is a sufficiently high concentration of protective colloids to cover the entire surface of all the unstable colloid particles. In fact, if gum Arabic addition is insufficient, it cannot prevent colloids precipitate. Preliminary laboratory trials to test the stabilizing effect of different dosages, with the help of the color stability test, are crucial to find the correct addition rate.
2) Bentonite fining
With its negative charge, bentonite can react with the positive unstable colloids involved in color precipitation (proteins and anthocyanins in the form of flavylium) and pull them down. Compared to the use of gum Arabic, bentonite fining is an operation that is more time and labor consuming, can decrease wine color and does not have a permanent effect: colloidal color matter is known to form regularly during aging and a wine may again be unstable at cold temperatures even a few months after fining. Nevertheless, bentonite can be used to reduce the instability of very color unstable wines and to prepare them for complete and long-lasting stabilization by addition of gum Arabic.